Ma Lab, Thomas Jefferson University
|
What are the mechanisms of dendritic branching?
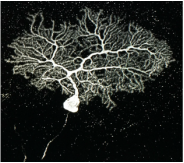
The Ma lab focuses on the branching of nerve cells. Though branching of axons and dendrites is common in every neuron cell type and a key step in establishing synaptic connections, the mechanism underlying their formation is not well understood.
Purkinje cells of the cerebellum exhibit some of the most elaborate dendritic branching seen in cells. We study these Purkinje cells using a viral delivery technique to inactivate gene function while simultaneously visualizing single cell morphology. The Ma lab has identified a cell-surface signaling system required for self-avoidance in the Purkinje cell. This signaling system allows branches of the same cell to recognize itself and avoid overlaps, thus helping to form much more sensitive circuitry systems.
Laboratory skills: molecular cloning, AAV genome editing, CRISPR/cas9 genome editing, immunolabeling, brain tissue clearing, in situ hybridization, confocal microscopy, polymerase chain reaction, Neurolucida, gait analysis, western blotting
The Ma lab focuses on the branching of nerve cells. Though branching of axons and dendrites is common in every neuron cell type and a key step in establishing synaptic connections, the mechanism underlying their formation is not well understood.
Purkinje cells of the cerebellum exhibit some of the most elaborate dendritic branching seen in cells. We study these Purkinje cells using a viral delivery technique to inactivate gene function while simultaneously visualizing single cell morphology. The Ma lab has identified a cell-surface signaling system required for self-avoidance in the Purkinje cell. This signaling system allows branches of the same cell to recognize itself and avoid overlaps, thus helping to form much more sensitive circuitry systems.
Laboratory skills: molecular cloning, AAV genome editing, CRISPR/cas9 genome editing, immunolabeling, brain tissue clearing, in situ hybridization, confocal microscopy, polymerase chain reaction, Neurolucida, gait analysis, western blotting
Hegg Lab, Michigan State University
What are the mechanisms of inflammation in the olfactory epithelium?
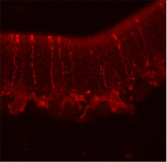
The Hegg lab studies mechanisms of neuroregeneration using the mouse olfactory epithelium (OE) as a model. Our noses incur damage on a daily basis, and the OE is able to regenerate and maintain function. However, if chronic inflammation is initiated, it can lead to olfactory dysfunction. I studied modulation of acute inflammation in the mouse OE to develop understanding of chronic inflammation. My central hypothesis was that environmental irritants activate inflammation by acting on trigeminal nerve receptors to release inflammatory neuropeptides. Neuropeptides then activate the release of cytokines from macrophages and initiate an inflammatory cascade. We expect our work will direct studies toward developing therapies for olfactory dysfunction and chronic inflammation.
Laboratory skills: confocal imaging, live cell imaging, tissue digestion and DNA extraction, polymerase chain reaction, gel electrophoresis, immunohistochemistry and immunofluorescence, enzyme-linked immunoassays, olfactory behavioral testing, cell culture, metamorph image analysis, transgenic animal colony management, small animal surgery, statistical analyses.
The Hegg lab studies mechanisms of neuroregeneration using the mouse olfactory epithelium (OE) as a model. Our noses incur damage on a daily basis, and the OE is able to regenerate and maintain function. However, if chronic inflammation is initiated, it can lead to olfactory dysfunction. I studied modulation of acute inflammation in the mouse OE to develop understanding of chronic inflammation. My central hypothesis was that environmental irritants activate inflammation by acting on trigeminal nerve receptors to release inflammatory neuropeptides. Neuropeptides then activate the release of cytokines from macrophages and initiate an inflammatory cascade. We expect our work will direct studies toward developing therapies for olfactory dysfunction and chronic inflammation.
Laboratory skills: confocal imaging, live cell imaging, tissue digestion and DNA extraction, polymerase chain reaction, gel electrophoresis, immunohistochemistry and immunofluorescence, enzyme-linked immunoassays, olfactory behavioral testing, cell culture, metamorph image analysis, transgenic animal colony management, small animal surgery, statistical analyses.
Byrd-Jacobs Lab, Western Michigan University
When sensory cells of the peripheral nervous system are lost, how is the central nervous system affected?
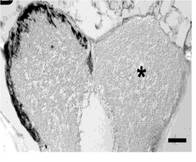
Dr. Byrd-Jacobs' research program focuses on plasticity of the adult central nervous system using the olfactory system of zebrafish as a model. We work to discover the cellular interactions and molecular factors responsible for the maintenance of brain structures and their ability to respond to injury. This research has important implications for neural regeneration and recovery from brain injury and disease. My project involved using repeated chemical-induced injury (ablation) of neurons of the olfactory epithelium in order to determine the timescale and phenotype of neuronal recovery in the olfactory epithelium. This repeated ablation not only kills mature neurons, but also degenerates the olfactory nerve, which deprives the olfactory bulb of sensory information. Thus, I also used this repeated ablation paradigm to examine how the bulb changed as it was separated from the olfactory epithelium, and whether allowing the olfactory epithelium time to recover would allow for the structure and neurochemistry of the bulb to improve given the return of sensory information to the olfactory bulb.
Laboratory skills: immunohistochemistry, histology, axonal tract tracing, small animal surgery, ImageJ morphometric analysis
Dr. Byrd-Jacobs' research program focuses on plasticity of the adult central nervous system using the olfactory system of zebrafish as a model. We work to discover the cellular interactions and molecular factors responsible for the maintenance of brain structures and their ability to respond to injury. This research has important implications for neural regeneration and recovery from brain injury and disease. My project involved using repeated chemical-induced injury (ablation) of neurons of the olfactory epithelium in order to determine the timescale and phenotype of neuronal recovery in the olfactory epithelium. This repeated ablation not only kills mature neurons, but also degenerates the olfactory nerve, which deprives the olfactory bulb of sensory information. Thus, I also used this repeated ablation paradigm to examine how the bulb changed as it was separated from the olfactory epithelium, and whether allowing the olfactory epithelium time to recover would allow for the structure and neurochemistry of the bulb to improve given the return of sensory information to the olfactory bulb.
Laboratory skills: immunohistochemistry, histology, axonal tract tracing, small animal surgery, ImageJ morphometric analysis
Pagano Lab, Henry Ford Health Systems
What is the role of adventitial vascular tissue in hypertension?
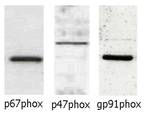
Dr. Pagano’s research examines the role of the adventitial layer in modulating the structure and function of vascular tissue, in both physiological and disease conditions. He was among the first to identify subunit p67phox in non-phagocytic NADPH oxidase of the vascular wall. He has gone on to demonstrate the activation of p67phox and NADPH-oxidase in response to the pro-hypertensive hormone angiotensin II. Importantly, Dr. Pagano developed an adenoviral inhibitor of NADPH oxidase, widely considered the most specific available. For my projects, we hypothesized that increasing the activity of NADPH-oxidase in adventitial fibroblasts with angiotensin II causes the release of hormones that increase hypertrophy of the media, and that these factors increase hypertrophy by activating the ERK1/2 pathway. We studied the major cell type of the adventitia, called fibroblasts, to gain a better understanding of how the adventitia might affect medial hypertrophy, using an NADPH oxidase activity assay, a proliferation assay, and Western blotting. We found that the proliferation of fibroblasts and secretion of cyclophilin-a in response to AngII were NADPH-oxidase dependent.
Laboratory skills: cell culture, western blotting, luminometry based activity assays
Dr. Pagano’s research examines the role of the adventitial layer in modulating the structure and function of vascular tissue, in both physiological and disease conditions. He was among the first to identify subunit p67phox in non-phagocytic NADPH oxidase of the vascular wall. He has gone on to demonstrate the activation of p67phox and NADPH-oxidase in response to the pro-hypertensive hormone angiotensin II. Importantly, Dr. Pagano developed an adenoviral inhibitor of NADPH oxidase, widely considered the most specific available. For my projects, we hypothesized that increasing the activity of NADPH-oxidase in adventitial fibroblasts with angiotensin II causes the release of hormones that increase hypertrophy of the media, and that these factors increase hypertrophy by activating the ERK1/2 pathway. We studied the major cell type of the adventitia, called fibroblasts, to gain a better understanding of how the adventitia might affect medial hypertrophy, using an NADPH oxidase activity assay, a proliferation assay, and Western blotting. We found that the proliferation of fibroblasts and secretion of cyclophilin-a in response to AngII were NADPH-oxidase dependent.
Laboratory skills: cell culture, western blotting, luminometry based activity assays
